Why Are Coagulant Chemicals Essential for Water Treatment Processes?
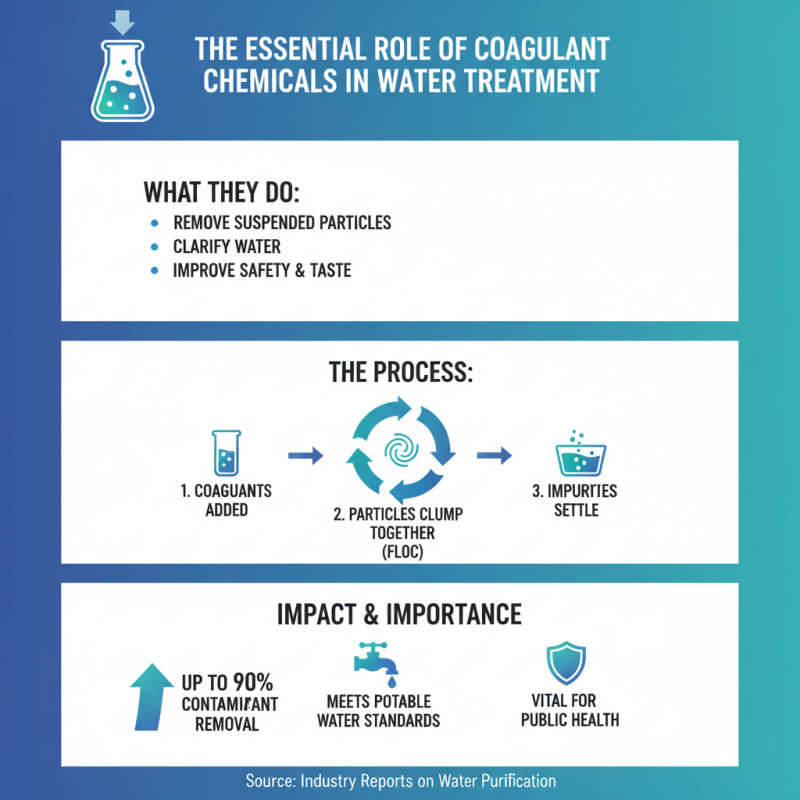
Water treatment plays a critical role in ensuring safe drinking water for communities worldwide. Coagulant chemicals are essential in this process. They help remove suspended solids and impurities from water. According to the World Health Organization, approximately 2 billion people lack access to safe drinking water. Effective coagulation can significantly reduce contaminants, improving water quality.
Studies show that using coagulant chemicals, such as aluminum sulfate and ferric chloride, enhances sedimentation and filtration. In fact, a report by the American Water Works Association highlights that coagulant chemicals can reduce turbidity by up to 95%. However, the choice of coagulant is not straightforward. It requires careful assessment of water quality and desired outcomes. Inefficient use can lead to poor water quality and health risks.
Moreover, the environmental impact of these chemicals must be considered. While coagulant chemicals are effective, they can also pose risks if not managed correctly. Ensuring optimal dosing and minimizing waste is crucial. Balancing efficacy with environmental considerations challenges water treatment professionals. Understanding these dynamics is vital to the future of water treatment strategies.

The Role of Coagulant Chemicals in Water Treatment
Coagulant chemicals play a pivotal role in water treatment. These substances help remove suspended particles and impurities from water. The process of coagulation aids in clarifying water, making it safer and more palatable. According to industry reports, effective coagulation can improve removal rates of contaminants by up to 90%. This is vital for meeting regulatory standards for potable water.
Using coagulants not only enhances water quality but also optimizes filtration processes. For example, aluminum sulfate is a common coagulant that can significantly reduce turbidity. The chemical forms larger particles, or flocs, that settle faster, ultimately saving energy and time in treatment facilities. However, reliance on chemical coagulants raises concerns about residuals in treated water.
Tips: Monitor the dosage of coagulants closely. Too much can create unwanted waste. Regular testing for residuals ensures safe water delivery. Adjustments based on current water quality are crucial for optimal results. A well-tuned approach can lead to improved efficiency and reduced chemical costs. Always consider the environmental impact of chemical use and explore sustainable alternatives when possible.
Types of Coagulant Chemicals Used in water Treatment
Coagulant chemicals play a vital role in water treatment processes. They help remove suspended particles and impurities from water, ensuring it is safe for consumption. Various types of coagulants are used in these processes, each with unique properties and applications.
One commonly used coagulant is aluminum sulfate, or alum. It is effective in aggregating small particles, making them easier to filter out. Research shows that alum can reduce turbidity levels significantly, often by as much as 90%. Another type is ferric chloride, which is known for its high efficiency in low pH conditions. Studies indicate that ferric chloride can enhance the removal of organic substances in water, contributing to improved quality.
Polymeric coagulants are gaining popularity for their flexibility and effectiveness. These coagulants can adjust to varying water compositions, thus providing consistent performance. However, their higher costs can lead to hesitancy in adoption. Water treatment facilities must balance effectiveness against economic considerations. Inadequate use of coagulants can lead to incomplete contaminant removal, impacting water quality. Regular monitoring and adjustments are crucial for optimal performance.
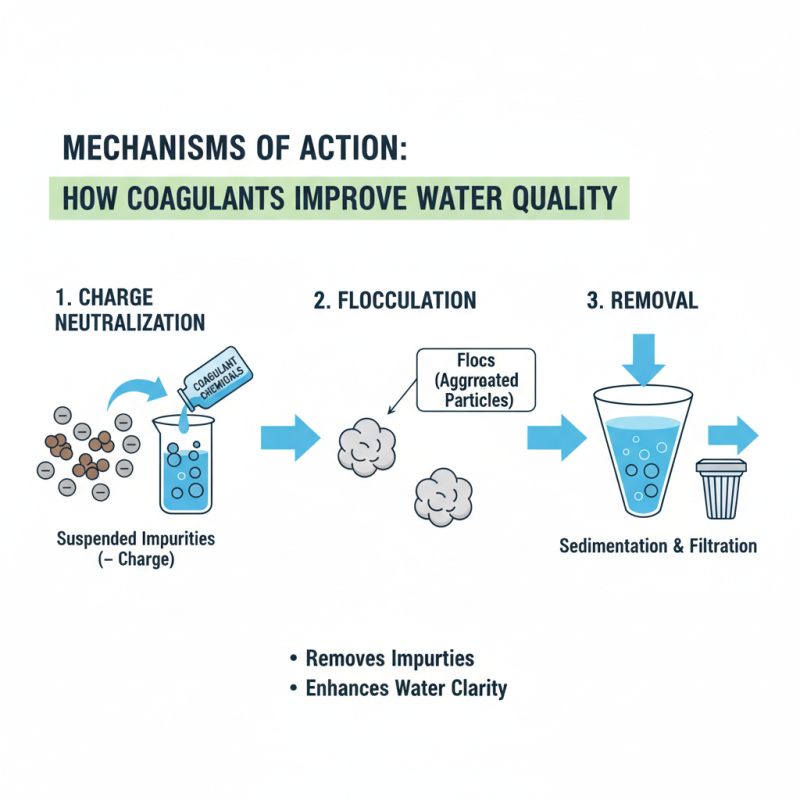
Mechanisms of Action: How Coagulants Improve Water Quality
Coagulant chemicals play a vital role in water treatment. They assist in removing impurities, enhancing water clarity. These substances work by neutralizing charges on suspended particles, facilitating their aggregation. When particles cluster together, they form larger entities, known as flocs. This process allows easier removal through sedimentation or filtration.
In water treatment facilities, common coagulants include aluminum sulfate and ferric chloride. Their application not only improves water quality but also reduces turbidity. When the water is clear, it is more visually appealing and safe for consumption.
**Tip:** Always monitor coagulant dosage. Too little may result in ineffective treatment. Too much can lead to excess chemical residues.
The effectiveness of coagulants can vary based on water quality. High organic loads may require adjustments in dosage. Operators need to be vigilant and adaptive. Testing water at different stages ensures the process remains efficient.
**Tip:** Regular testing helps identify changes in water characteristics. This proactive approach aids in selecting the right coagulant and dosage.
Proper understanding of these mechanisms can significantly enhance water treatment outcomes. Continual learning in this field is essential for improvement and adaptability.
Impact of Coagulant Selection on Treatment Efficiency and Costs
The selection of coagulant chemicals can significantly influence the efficiency of water treatment processes. Different coagulants interact with impurities in various ways. A poorly chosen coagulant may not effectively remove suspended particles or lead to suboptimal floc formation. This can result in higher levels of contaminants remaining in treated water, posing health risks.
In terms of costs, the choice of coagulant also plays a critical role. Some coagulants may require additional processing steps or post-treatment adjustments. This can increase operational expenses. Conversely, an effective coagulant can enhance sedimentation rates, leading to reduced energy and time costs. However, this might come with a higher upfront cost.
Understanding the water source and treatment goals is essential for making informed decisions. Not all treatment facilities have the same needs. Operator expertise also affects how well a coagulant performs. Continuous evaluation and adjustment of coagulant selection are necessary. This reflection helps maintain both efficiency and cost-effectiveness in water treatment practices.
Impact of Coagulant Selection on Water Treatment Efficiency
Environmental Considerations in the Use of Coagulants in Water Treatment
Coagulants play a pivotal role in water treatment by improving water clarity. However, their environmental impact requires careful consideration. Many chemical coagulants, while effective, can contribute to ecological risks. For instance, aluminum sulfate can accumulate in aquatic systems, potentially harming fish and other wildlife.
The sourcing of coagulant chemicals can also pose ethical questions. Some industries may prioritize cost over environmental sustainability. Practices like mining for aluminum can lead to habitat destruction and pollution. Ensuring that coagulants are sourced responsibly is vital for safeguarding ecosystems.
Moreover, the residuals from coagulants need attention. After treatment, these substances can linger in our waterways. This residue could affect water quality and ecosystem health. It is essential for water treatment facilities to adopt strategies to minimize these impacts. They must strike a balance between effective water purification and environmental stewardship. Ongoing research into biodegradable alternatives might pave the way for greener practices in the future.
Why Are Coagulant Chemicals Essential for Water Treatment Processes? - Environmental Considerations in the Use of Coagulants in Water Treatment
| Coagulant Chemical |
Type |
Applications |
Environmental Impact |
| Aluminum Sulfate |
Inorganic Coagulant |
Water treatment for drinking and wastewater |
Can cause aluminum accumulation in water bodies |
| Ferric Chloride |
Inorganic Coagulant |
Used in municipal and industrial wastewater treatment |
Can lead to iron deposits in aquatic systems |
| Polymeric Coagulants |
Synthetic Coagulant |
Sedimentation and clarification processes |
Lower environmental impact; biodegradable options available |
| Calcium Hydroxide |
Inorganic Coagulant |
pH adjustment and softening |
Potential contribution to high pH levels in effluents |