2026 How to Choose the Right Coagulant Chemical for Water Treatment?
Choosing the right coagulant chemical for water treatment is crucial for efficiency. Dr. John Smith, a renowned expert in water management, emphasizes, "The effectiveness of water treatment hinges on the right coagulant." Coagulant chemicals play a pivotal role in clarifying water. They help to remove impurities and suspended particles, making water safe for use.
Navigating the vast array of available coagulant chemicals can be overwhelming. Factors like water quality, treatment goals, and cost come into play. Not all coagulants are created equal; each has specific strengths and weaknesses. For example, aluminum sulfate is commonly used, but it might not be the best choice for every situation.
Understanding the characteristics of coagulant chemicals requires careful consideration. Achieving the ideal balance often demands trial and error. The chosen coagulant may perform well but might not be the most cost-effective. This highlights the importance of reflection in decision-making for water treatment processes.

Understanding the Role of Coagulants in Water Treatment Processes
Coagulants play a critical role in water treatment processes. These chemicals help to remove suspended particles, making water clearer and safer. By neutralizing the charge on particles, coagulants allow them to clump together. This process forms larger aggregates called flocs, which can then be easily removed.
Choosing the right coagulant requires understanding water chemistry. Different sources of water may need specific types of coagulants. For example, turbid river water may require different treatment than clear lake water. Testing water quality is essential. This helps identify the best coagulant for effective treatment.
**Tips:** Always monitor water characteristics. Regular testing ensures the coagulant performs optimally. Consider environment-friendly options. Sometimes, less is more effective than a high dosage. Balancing treatment effectiveness with environmental impact is vital.
2026 How to Choose the Right Coagulant Chemical for Water Treatment? - Understanding the Role of Coagulants in Water Treatment Processes
| Coagulant Type |
Chemical Formula |
Optimal pH Range |
Usage |
Advantages |
Potential Issues |
| Alum |
KAl(SO₄)₂·12H₂O |
6.0 - 8.0 |
Drinking Water |
Cost-effective, widely used |
Can lower pH, residual aluminum |
| Ferric Chloride |
FeCl₃ |
4.0 - 6.5 |
Wastewater Treatment |
Effective in low pH, reduces sludge volume |
High iron content, stains |
| Polymeric Coagulants |
Varies |
6.5 - 8.5 |
Surface Water |
High efficiency, low dosage |
Cost, may require adjustment |
| Lime |
Ca(OH)₂ |
10 - 12 |
Hard Water Treatment |
Increases alkalinity, reduces hardness |
Requires careful handling |
| Soda Ash |
Na₂CO₃ |
8.0 - 10.0 |
pH Adjustment |
Increases pH, improves coagulation |
Can increase alkalinity |
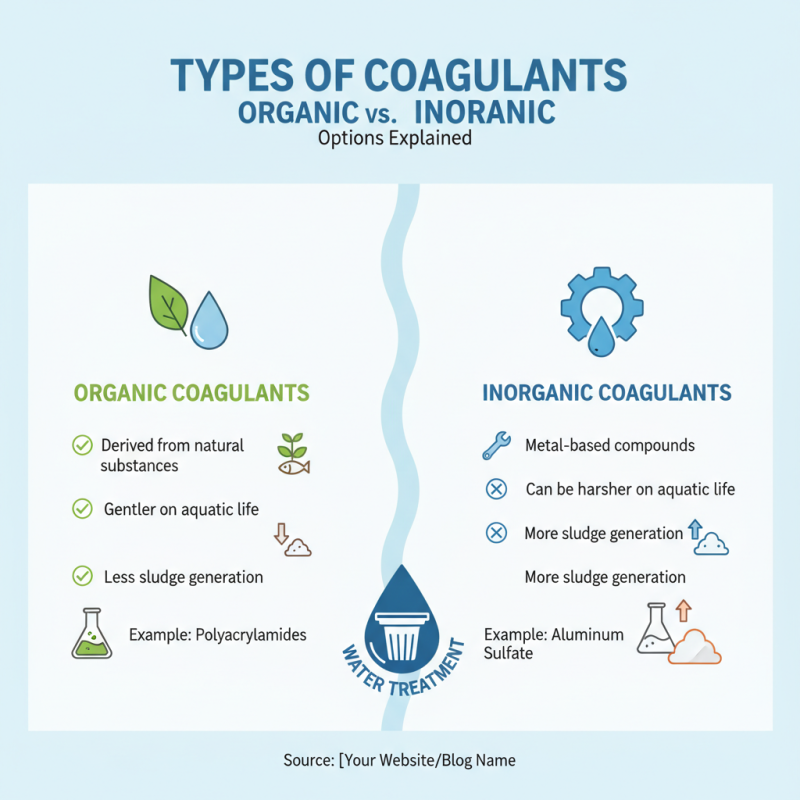
Types of Coagulants: Organic vs. Inorganic Options Explained
When selecting coagulants for water treatment, understanding the differences between organic and inorganic options is crucial. Organic coagulants, typically derived from natural substances, offer a gentler touch. They are less harsh on aquatic life. Yet, their performance can vary significantly. Some studies suggest they are effective in smaller dosages, leading to less sludge generation. An example is polyacrylamides, which enhance the settling rate of particles.
In contrast, inorganic coagulants, like aluminum sulfate and ferric chloride, have a long-standing history in water treatment. They tend to be more effective at removing turbidity. A report from the American Water Works Association indicates that over 70% of U.S. water treatment facilities utilize inorganic coagulants. However, they can produce more sludge, requiring additional processing. This trade-off presents a dilemma for water treatment plant operators. Each choice has implications for both efficiency and environmental impact.
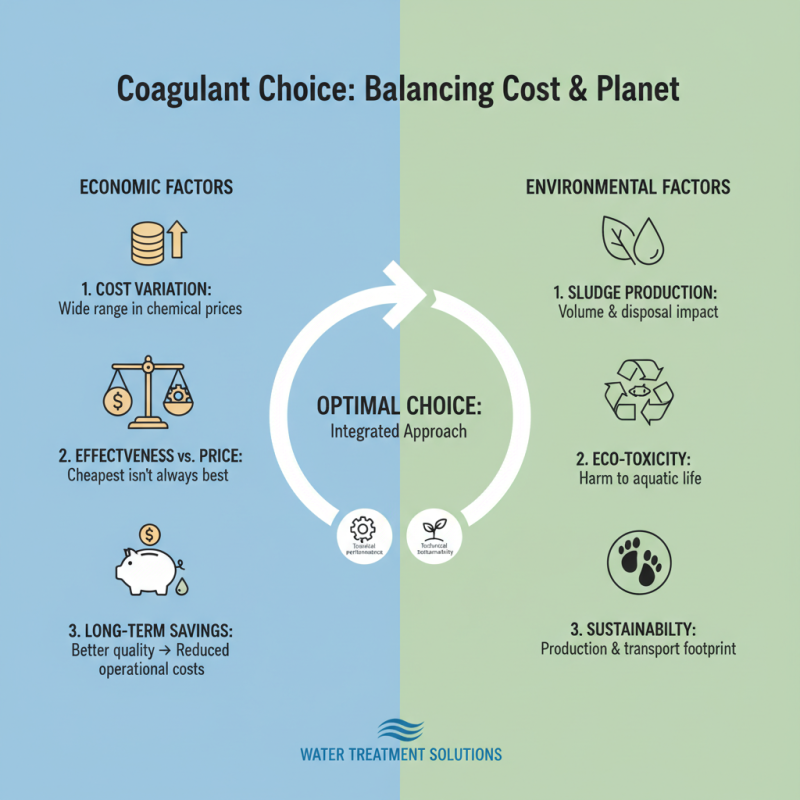
Understanding these nuances is vital for improving water treatment processes. Misjudgments in selection can lead to increased costs. They can also affect the quality of treated water. Balancing efficacy and environmental concerns is an ongoing challenge in the industry.
Evaluating Coagulant Efficiency: Key Performance Metrics and Industry Standards
Choosing the right coagulant for water treatment involves understanding its efficiency. Key performance metrics are essential for evaluation. These metrics include turbidity removal, sedimentation rates, and residual concentrations. Each coagulant has strengths and weaknesses. Therefore, it is crucial to assess their performance against industry standards.
When evaluating a coagulant's efficiency, consider testing under various conditions. Different water sources can alter results significantly. An effective coagulant in one region may fail elsewhere. Systematic testing helps identify the right fit.
**Tip:** Always keep a record of your findings for future reference.
Inconsistent performance may arise from changes in water quality. Adjustments in dosage and application methods can improve results. Regular monitoring is necessary to maintain optimal performance. Making informed decisions about coagulants can lead to better water quality outcomes.
**Tip:** Collaborate with experts for more precise evaluations.
Factors Influencing Coagulant Selection: Water Quality and Treatment Goals
Choosing the right coagulant for water treatment is crucial. Water quality varies greatly, influencing coagulant effectiveness. For instance, turbidity levels can range between 1 to 100 NTU or more. This variance emphasizes the need for specific coagulants based on the water's characteristics.
Alkalinity and pH are vital factors in coagulant selection. High alkalinity can foster optimal coagulation processes. Research from the Water Research Foundation suggests that a pH range of 6.5 to 8.5 maximizes efficiency. However, many operators overlook these parameters. A missed adjustment could lead to inefficiency in removal rates.
Treatment goals also dictate coagulant choice. Different objectives may require unique coagulant properties. If the goal is to reduce color or taste, selecting a coagulant with suitable charge density is essential. However, some plants rely on outdated data, risking suboptimal performance. Recognizing these gaps is critical for improving overall water treatment effectiveness.
Coagulant Chemical Selection for Water Treatment